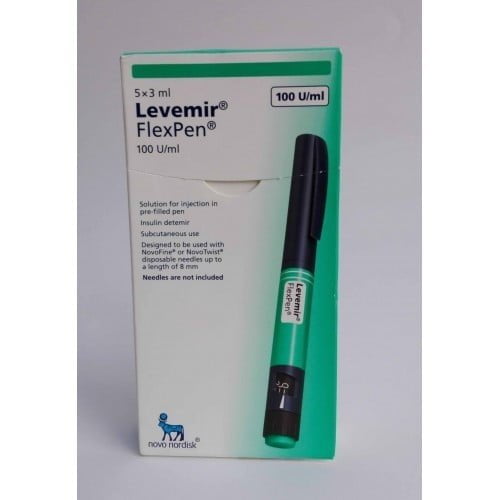
Levemir FlexPen 100iu/ml 3ml 1`s
2,000KSh
Categories PHARMACY ONLY, Anti-Diabetics
Share On
Product Description
- Levemir® is not recommended for the treatment of diabetic ketoacidosis.
- Levemir® is contraindicated during episodes of hypoglycemia and in patients hypersensitive to Levemir® or any of its excipients. Reactions have included anaphylaxis.
- Never Share a Levemir® FlexTouch® Pen, Needle, or Syringe Between Patients
- Hyperglycemia or Hypoglycemia with Changes in Insulin Regimen: Changes in an insulin regimen may affect glycemic control and predispose to hypoglycemia or hyperglycemia.
- Hypoglycemia: Hypoglycemia is the most common adverse reaction of insulin, including Levemir®, and may be life-threatening.
- Hypersensitivity and Allergic Reactions: If hypersensitivity reactions occur, discontinue Levemir®; treat per standard of care and monitor until symptoms and signs resolve. Levemir® is contraindicated in patients who have had hypersensitivity reactions to insulin detemir or any of the excipients.
- Hypokalemia: All insulin products, including Levemir®, cause a shift in potassium from the extracellular to intracellular space, possibly leading to hypokalemia.
- Fluid Retention and Heart Failure with Concomitant Use of PPAR-gamma Agonists: Thiazolidinediones (TZDs), which are peroxisome proliferator-activated receptor (PPAR)-gamma agonists, can cause dose-related fluid retention, particularly when used in combination with insulin